|
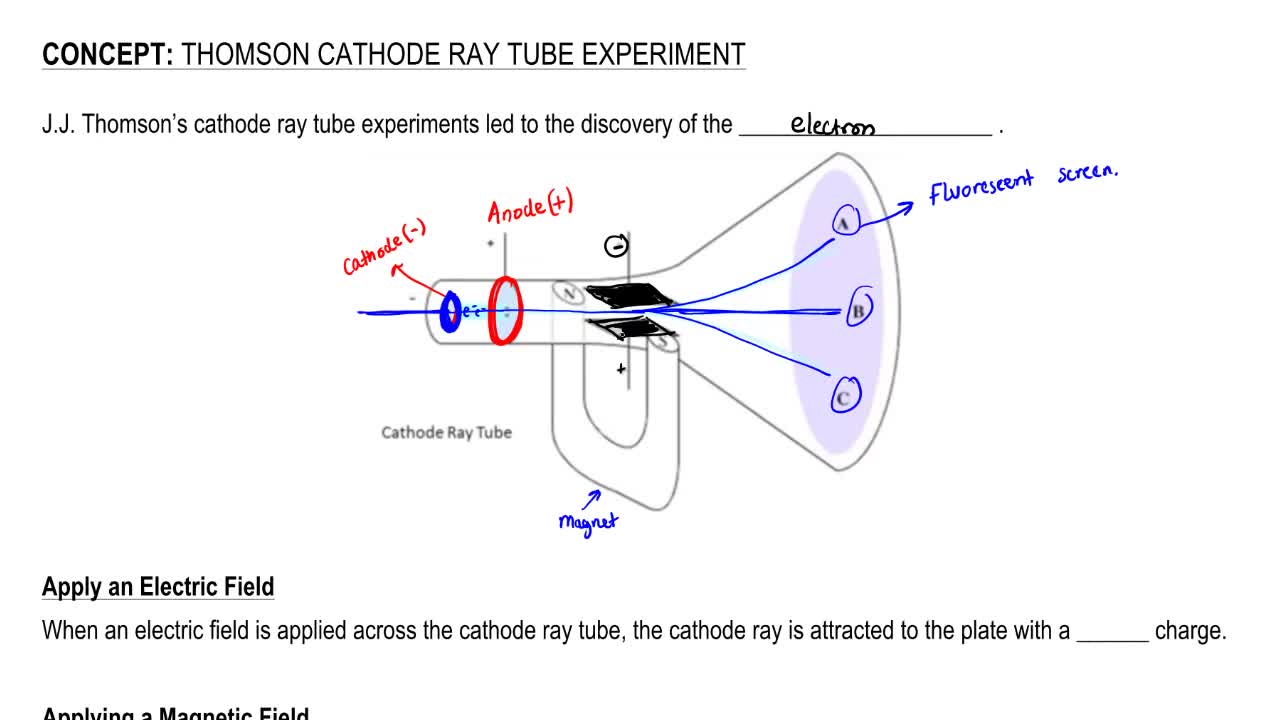
So much so, that George Stoney (1891) proposed the name electron for the particle. Scientist had believed in the existence of a negative particle for some time. Thomson demonstrated the existence of the electron. Their work culminated in the discovery by English physicist J.J. The cathode ray tube experiment performed by J.J. These subatomic particles can be found within atoms of all elements.Ĭonclusion - The cathode rays are made up of negatively charged particles known as electrons. During the 1880s and ’90s scientists searched cathode rays for the carrier of the electrical properties in matter. The ray is detected by painting a special coating of phosphorus. He had already found a similar value in his. The tubes are called cathode ray tubes because the particle beam originates at the cathode. 
In addition, the experiment could describe characteristic properties, in essence, its affinity to positive charge, and its charge to mass ratio. Thomson, is one of the most well-known physical experiments that led to electron discovery. He advanced the idea that cathode rays are really streams of very small pieces of atoms. Thomson’s experiments with cathode rays led to the discovery of the electron, the first subatomic particle to be discovered. JJ Thomson found the mass-to-charge ratio of the cathode rays was only about 1/2000 of that of hydrogen nuclei. The experiment Cathode Ray Tube (CRT) conducted by J. Thomson also placed two magnets on either side of the tube, and observed that this magnetic field also deflected the cathode ray. In the year 1897, the English physicist J.J. This indicated that the cathode ray was composed of negatively-charged particles. The cathode ray was deflected away from the negatively-charged electric plate and towards the positively-charged plate. Thomson announced the results of his previous four months experiments on cathode rays. Thomson's experiment on constituents of cathode rays:Įxperiment - To test the properties of the particles, Thomson placed two oppositely-charged electric plates around the cathode ray. An electric discharge is passed through the tube.Įmission of blue rays can be seen from the cathode.Ĭonclusion - The blue rays emitted from cathode are cathode rays. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
